U.S. Cell and Gene Therapy CDMO Market is projected to grow from USD 2.09 Billion in 2025 to USD 10.28 Billion by 2035 due to expanding gene and cell therapy pipelines globally
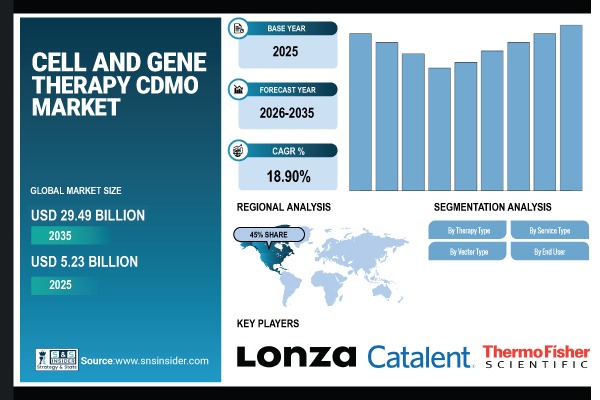
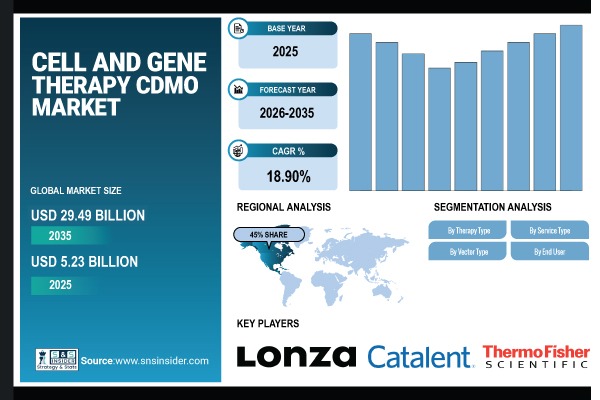
Austin, March 06, 2026 (GLOBE NEWSWIRE) — According to SNS Insider, The Cell and Gene Therapy CDMO Market size was valued at USD 5.23 Billion in 2025 and is expected to reach USD 29.49 Billion by 2035, growing at a CAGR 18.90% during 2026-2035.
The Cell and Gene Therapy CDMO market is expanding rapidly due to the rising number of advanced therapies for oncology, rare diseases, and genetic disorders. Growing manufacturing complexity, high costs, and strict regulatory requirements are driving biopharma companies to outsource development and production to CDMOs. These organizations provide capabilities in process development, viral vector production, cell processing, and commercial manufacturing. Increasing capacity, technological advancements, and regulatory support are further strengthening the role of CDMOs in accelerating clinical development and commercialization of cell and gene therapies.
Cell and Gene Therapy CDMO Market Size and Forecast
- Market Size in 2025: USD 5.23 Billion
- Market Size by 2035: USD 29.49 Billion
- CAGR: 18.90%
- Base Year: 2025
- Forecast Period: 2026-2035
- Historical Data: 2022-2024
Request Instant Access to the Cell and Gene Therapy CDMO Market Forecast @ https://www.snsinsider.com/sample-request/9749
The U.S. Cell and Gene Therapy CDMO Market is projected to grow from USD 2.09 Billion in 2025 to USD 10.28 Billion by 2035, at a CAGR of 17.26%.
Growth is driven by expanding cell and gene therapy pipelines, strong FDA regulatory support, increasing outsourcing by biopharma companies, rising viral vector demand, and continuous investments in advanced manufacturing infrastructure.
Increased Cell & Gene Therapy Pipeline / Outsourced Demand Augment Market Growth Globally
The market is driven by an increasing number of commercial and clinical cell and gene therapy initiatives for genetic disorders, uncommon diseases, and cancer. However, biopharma producers are well aware of the necessity for a CDMO: sophisticated manufacturing, capital-intensive facility investments, and stringent regulatory requirements are pushing businesses to contract with specialized CDMOs for both development and production. Market expansion is also facilitated by regulatory incentives, such as the FDA RMAT and expedited approval processes, which enable quicker clinical translation and commercialization.
Cell and Gene Therapy CDMO Market Segmentation Analysis:
By Therapy Type
In 2025, Cell Therapy dominated with 49% share due to the high volume of clinical-stage and commercial programs, particularly CAR-T, TCR, and stem-cell-based therapies. Gene therapy is the fastest growing modality propelled by a vast growing pipeline for AAV and lentiviral products for rare genetic, neurological, and inherited metabolic disorders.
By Service Type
In 2025, Manufacturing dominated with 57% share due to its capital-intensive nature, high technical complexity, and strict regulatory requirements. Process and analytical development is the largest and fastest growing service segment owing to a growing early-stage CGT pipeline as well as requirement of process that are scalable, optimized and reproducible.
By Vector Type
In 2025, Viral Vectors dominated with 69% share due to their high transduction efficiency, established clinical performance, and broad regulatory acceptance. Non-viral vectors are the fastest growing sector, driven by developments in lipid nanoparticles, plasmid DNA and gene-editing delivery technologies.
By End-User
In 2025, Biopharmaceutical Companies dominated with 61% share due to the large number of CGT pipeline, huge financial resources and dependence on CDMOs for multi-program development and commercial manufacturing. Startups represent the fastest-growing end-user segment as virtual biotech models, increased venture funding, and academic spin-outs continue to expand the CGT ecosystem.
For Analyst-Curated Insights Customized to Your Business, Connect Now @ https://www.snsinsider.com/request-analyst/9749
Cell and Gene Therapy CDMO Market Regional Insights:
North America dominates the global Cell and Gene Therapy CDMO market, accounting for approximately 45% of total market revenue. This leadership is supported by a high density of biotech and pharmaceutical companies and a large, advanced cell and gene therapy clinical development pipeline, along with strong regulatory support including from the FDA.
Asia-Pacific is the fastest-growing region in the Cell and Gene Therapy CDMO market, registering a CAGR of 23.05% during 2026-2035. Rapid developments are spurred on by increasing biomanufacturing capacity, the cost-effectiveness of production and growing government support for advanced therapeutics.
Major Players Analysis Listed in the Cell and Gene Therapy CDMO Market Report:
- Lonza Group
- Catalent, Inc.
- Thermo Fisher Scientific
- WuXi Advanced Therapies
- Samsung Biologics
- Charles River Laboratories
- AGC Biologics
- Oxford Biomedica
- Fujifilm Diosynth Biotechnologies
- Viralgen Vector Core
- Kite Pharma Manufacturing
- Resilience
- Bayer Cell Therapy Manufacturing
- Yposkesi
- KBI Biopharma
- Brammer Bio
- Cobra Biologics
- Cell and Gene Therapy Catapult Manufacturing Innovation Centre
- Porton Advanced Solutions
- Novasep
Cell and Gene Therapy CDMO Market Recent Developments:
- In February 2025: Lonza expanded its cell and gene therapy manufacturing capacity in the United States to support increasing demand for late-stage and commercial CGT programs, strengthening its global CDMO footprint.
- In January 2025: Catalent strengthened its viral vector manufacturing capabilities through facility upgrades and technology investments aimed at supporting high-volume gene therapy programs.
Purchase Single User PDF of Cell and Gene Therapy CDMO Market Report @ https://www.snsinsider.com/checkout/9749
Exclusive Sections of the Report (The USPs):
- CGT OUTSOURCING & ADOPTION METRICS – helps you understand the percentage of organizations outsourcing cell and gene therapy manufacturing for core and experimental programs, along with the annual growth in CDMO engagement across therapy types.
- THERAPY-SPECIFIC MANUFACTURING TRENDS – helps you analyze adoption patterns across cell therapy, gene therapy, and gene-modified cell therapy manufacturing, highlighting areas with the fastest growth in outsourced production.
- ADVANCED MANUFACTURING TECHNOLOGY ADOPTION – helps you evaluate the shift toward automated, closed-system, and single-use manufacturing platforms, as well as the share of viral, non-viral, and cell-based delivery production methods.
- PERFORMANCE, COST & REGULATORY COMPLIANCE METRICS – helps you assess improvements in manufacturing efficiency, batch success rates, compliance with GMP and regulatory standards, and the cost advantages of outsourcing versus in-house production.
- CDMO FACILITY UTILIZATION & PROGRAM SUPPORT – helps you understand the number of CGT programs supported per CDMO facility annually and the level of service adoption across different therapy development stages.
- SPONSOR VALUE CREATION & PARTNERSHIP INSIGHTS – helps you gauge sponsor satisfaction, ROI outcomes, and long-term partnership retention through customized, therapy-specific manufacturing solutions.
Cell and Gene Therapy CDMO Market Report Scope:
| Report Attributes | Details |
| Market Size in 2025 | USD 5.23 Billion |
| Market Size by 2035 | USD 29.49 Billion |
| CAGR | CAGR of 18.90% From 2026 to 2035 |
| Base Year | 2025 |
| Forecast Period | 2026-2035 |
| Historical Data | 2022-2024 |
| Key Segments | • By Therapy Type: (Cell Therapy, Gene Therapy, Gene-Modified Cell Therapy) • By Service Type: (Process & Analytical Development, Manufacturing, Fill & Finish Services) • By Vector Type: (Viral Vectors, Non-Viral Vectors, Cell-Based Delivery Systems) • By End User: (Biopharmaceutical Companies, Biotechnology Startups, Academic & Research Institutes) |
| Regional Analysis/Coverage | North America (US, Canada), Europe (Germany, UK, France, Italy, Spain, Russia, Poland, Rest of Europe), Asia Pacific (China, India, Japan, South Korea, Australia, ASEAN Countries, Rest of Asia Pacific), Middle East & Africa (UAE, Saudi Arabia, Qatar, South Africa, Rest of Middle East & Africa), Latin America (Brazil, Argentina, Mexico, Colombia, Rest of Latin America). |
Rising Demand for Cell and Gene Therapy CDMO Market Data, Our Full Report Trend Analysis @ https://www.snsinsider.com/reports/cell-and-gene-therapy-cdmo-market-9749
About Us:
SNS Insider is one of the leading market research and consulting agencies that dominates the market research industry globally. Our company’s aim is to give clients the knowledge they require in order to function in changing circumstances. In order to give you current, accurate market data, consumer insights, and opinions so that you can make decisions with confidence, we employ a variety of techniques, including surveys, video talks, and focus groups around the world.
CONTACT: Contact Data Contact Us: Rohan Jadhav - Principal Consultant Phone: +1-315 636 4242 (US) | +44- 20 3290 5010 (UK)

Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaShorts takes no editorial responsibility for the same.