Europe in vitro diagnostics (IVD) market is experiencing significant growth, driven by an aging population, rising chronic diseases, and advancements in molecular diagnostics. Key growth factors include high demand for personalized medicine, increased investment in AI, and a strong shift towards home-based testing, and the “Big Five” nations (Germany, UK, France, Italy, Spain) dominating the market.
Chicago, Feb. 06, 2026 (GLOBE NEWSWIRE) — According to recent data from Astute Analytica, the Europe in vitro diagnostics market was valued at US$ 24.85 billion in 2023 and is projected to attain a market size of US$ 36.93 billion by 2032 at a CAGR of 4.5% during the forecast period 2024–2032.
Europe is distinguished by one of the oldest populations in the world, a demographic trend that is shaping the region’s healthcare landscape in profound ways. According to the World Health Organization, by the year 2050, nearly 40% of Europeans will be aged 60 or older. This dramatic shift towards an aging society presents both challenges and opportunities, influencing healthcare priorities, resource allocation, and the demand for specialized medical services.
Request Sample Pages: https://www.astuteanalytica.com/request-sample/europe-in-vitro-diagnostics-market
The aging demographic in Europe is closely linked to a growing prevalence of chronic diseases, which tend to increase with age. Conditions such as diabetes, cardiovascular diseases, and various forms of cancer are becoming increasingly common, placing significant strain on healthcare systems. These chronic illnesses require long-term management and often involve complex treatment regimens, highlighting the critical need for effective diagnostic, therapeutic, and preventive strategies tailored to the elderly population.
Europe: A Hub of Innovation and Opportunity in In-Vitro Diagnostics
Europe stands as a vibrant melting pot of culture, innovation, and medical advancements, making it one of the most lucrative and dynamic markets for In Vitro Diagnostics (IVD) worldwide. The region’s rich history of scientific research, combined with its commitment to high-quality healthcare, has fostered an environment where diagnostic technologies can thrive.
At the heart of Europe’s IVD market are five powerhouse countries often referred to as the “Big Five”: the United Kingdom, Germany, France, Italy, and Spain. These nations collectively dominate the market, together accounting for an impressive 70% of the total European IVD market share. Their leadership is a reflection of strong healthcare systems, substantial research and development activities, and large patient populations requiring advanced diagnostic services.
Among the Big Five, Germany stands out as the undisputed leader in the European IVD market. This dominant position is driven by the country’s thriving healthcare infrastructure, which supports widespread access to advanced diagnostic technologies. Germany’s healthcare system emphasizes preventative medicine and early disease detection, fueling demand for cutting-edge IVD tools.
Decline of “One-Size-Fits-All” Medicine: Embracing Personalization
The era of “one-size-fits-all” medicine is gradually giving way to a more nuanced and patient-centric approach known as personalized or precision medicine. This transformative shift, gaining significant momentum within the European medical community, reflects a fundamental change in how healthcare is delivered. Instead of applying uniform treatments across broad patient groups, personalized medicine seeks to tailor therapies to the unique genetic, environmental, and lifestyle factors of each individual.
Central to the rise of personalized medicine is the powerful technology of genomic profiling. By decoding an individual’s genetic makeup, clinicians gain invaluable insights into disease risks, progression, and treatment responses. This molecular-level understanding enables customized therapeutic strategies that target the underlying causes of illness, rather than just managing symptoms.
The European Commission has recognized the immense potential of personalized medicine, investing over €1 billion through its Horizon 2020 programme to accelerate research and development in this field. These substantial investments have catalyzed advancements in diagnostic technologies, drug development, and clinical applications. By fostering collaboration between research institutions, industry, and healthcare providers, the programme has helped translate scientific discoveries into practical tools that benefit patients across Europe.
Immunodiagnostics: The Current Market Leader
Immunodiagnostics presently holds the dominant position in the diagnostics market, thanks to its widespread applications and proven effectiveness in detecting a variety of diseases through immune response markers. This segment has established itself as a cornerstone in clinical diagnostics, offering reliable, cost-effective, and rapid testing solutions. Its extensive use in detecting infections, autoimmune diseases, and allergies ensures sustained demand, reinforcing immunodiagnostics as a mainstay in healthcare institutions worldwide.
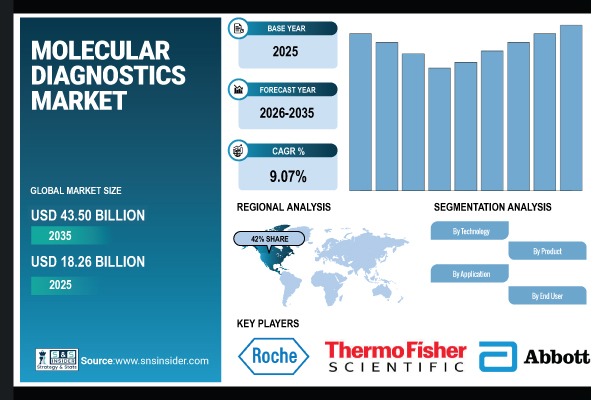
While immunodiagnostics leads today, it is the molecular diagnostics segment that exhibits remarkable potential for future expansion. Projected to achieve the highest compound annual growth rate (CAGR) of 4.9%, molecular diagnostics is rapidly climbing the ranks due to its transformative capabilities. This segment’s growth reflects increasing adoption of advanced molecular technologies that offer unparalleled precision and sensitivity, enabling clinicians to detect diseases at their earliest and most fundamental molecular stages.
The rising prominence of molecular diagnostics is largely attributed to its ability to analyze genetic material—DNA and RNA—offering a granular view of disease mechanisms that traditional diagnostics cannot match. This precision allows for early identification of infectious agents, genetic mutations, and biomarkers associated with various conditions.
Hospitals: The Cornerstones of the In-Vitro Diagnostics Market
Hospitals stand out as the undisputed leaders in the in-vitro diagnostics (IVD) market, commanding a substantial 41% share of total revenue. This dominant position is a direct reflection of their comprehensive infrastructure, which enables them to perform a wide array of diagnostic tests essential for patient care. As primary centers for advanced medical services, hospitals are uniquely equipped with the sophisticated laboratory facilities, skilled personnel, and technological resources required to conduct extensive diagnostic procedures efficiently and accurately.
A key factor contributing to hospitals’ leadership in the IVD market is the vast inflow of patients they manage daily. Hospitals cater to diverse patient populations, including those with complex and acute medical conditions that demand rapid and precise diagnostic results. Timely diagnostics are critical in guiding treatment decisions, improving patient outcomes, and streamlining care pathways.
The dominance of hospitals in the IVD market is further reinforced by the healthcare delivery models prevalent in many European countries. In these regions, centralized healthcare systems designate hospitals as primary care providers and diagnostic hubs, consolidating a significant portion of testing and clinical services within these institutions.
Europe In-Vitro Diagnostics Market Key Players
- Abbott
- Agilent Technologies, Inc.
- Becton Dickinson and Company
- bioMérieux SA
- Bio-Rad Laboratories, Inc.
- Charles River Laboratories
- Danaher Corporation
- F. Hoffmann-La Roche Ltd.
- Qiagen
- Quest Diagnostics
- Quidel Corp.
- Siemens Healthineers
- Sysmex Corp.
- Other Prominent Players
Key Segmentation:
By Product & Services
- Reagents
- Instruments
- Software
- Services
By Technique
- Immunodiagnostics
- Hematology
- Molecular Diagnostics
- Tissue Diagnostics
- Clinical Chemistry
- Others
By Application
- Cancer diagnostics
- Blood glucose monitoring
- Human genetic testing
- Immunoassays
- Hepatitis tests
- Infectious Diseases diagnostics
- Cardiac Diseases
- Nephrological Diseases
- Gastrointestinal Diseases
- Others
By End User
- Standalone Laboratories
- Hospitals
- Academic And Medical Schools
- Point Of Care
- Others
For more information about this report visit: https://www.astuteanalytica.com/industry-report/europe-in-vitro-diagnostics-market
About Astute Analytica
Astute Analytica is a global market research and advisory firm providing data-driven insights across industries such as technology, healthcare, chemicals, semiconductors, FMCG, and more. We publish multiple reports daily, equipping businesses with the intelligence they need to navigate market trends, emerging opportunities, competitive landscapes, and technological advancements.
With a team of experienced business analysts, economists, and industry experts, we deliver accurate, in-depth, and actionable research tailored to meet the strategic needs of our clients. At Astute Analytica, our clients come first, and we are committed to delivering cost-effective, high-value research solutions that drive success in an evolving marketplace.
Contact Us:
Astute Analytica
Phone: +1-888 429 6757 (US Toll Free); +91-0120- 4483891 (Rest of the World)
For Sales Enquiries: sales@astuteanalytica.com
Website: https://www.astuteanalytica.com/
Follow us on: LinkedIn | Twitter | YouTube
CONTACT: Contact Us: Astute Analytica Phone: +1-888 429 6757 (US Toll Free); +91-0120- 4483891 (Rest of the World) For Sales Enquiries: sales@astuteanalytica.com Website: https://www.astuteanalytica.com/

Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. IndiaShorts takes no editorial responsibility for the same.