The hepatic encephalopathy market is expected to witness steady growth over the coming years, driven by the rising prevalence of chronic liver diseases, cirrhosis, and alcohol-related liver disorders worldwide. Increasing awareness about early diagnosis and improved treatment options are further supporting market expansion. Additionally, the launch of emerging therapies such as VE303 (Vedanta Biosciences), YAQ007 (Yaqrit Discovery Ltd), 25% IV albumin (Grifols, S.A.), EBX-102 (EnteroBiotix Limited), GR3027 (Umecrine Cognition AB), and others will further propel the market growth.
New York, USA, May 04, 2026 (GLOBE NEWSWIRE) — Hepatic Encephalopathy Market Set for Growth at a 1.2% CAGR Through 2036, Driven by Emerging Therapies and Rising Disease Burden | DelveInsight
The hepatic encephalopathy market is expected to witness steady growth over the coming years, driven by the rising prevalence of chronic liver diseases, cirrhosis, and alcohol-related liver disorders worldwide. Increasing awareness about early diagnosis and improved treatment options are further supporting market expansion. Additionally, the launch of emerging therapies such as VE303 (Vedanta Biosciences), YAQ007 (Yaqrit Discovery Ltd), 25% IV albumin (Grifols, S.A.), EBX-102 (EnteroBiotix Limited), GR3027 (Umecrine Cognition AB), and others will further propel the market growth.
Recently published Hepatic Encephalopathy Market Insights report includes a comprehensive understanding of current treatment practices, hepatic encephalopathy emerging drugs, market share of individual therapies, and current and forecasted market size from 2022 to 2036, segmented into leading markets [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
Hepatic Encephalopathy Market Summary
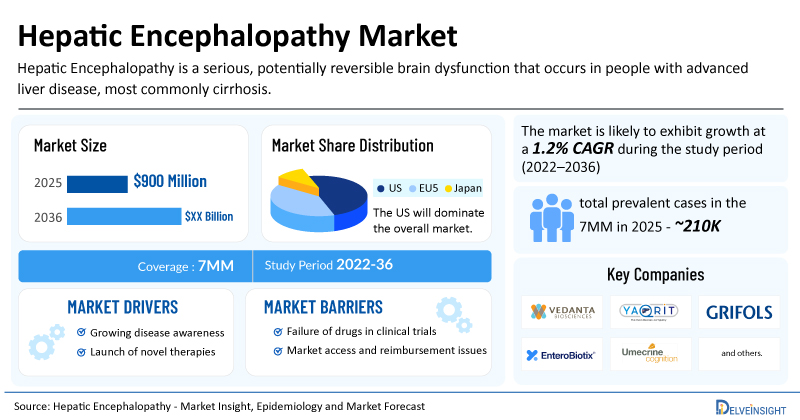
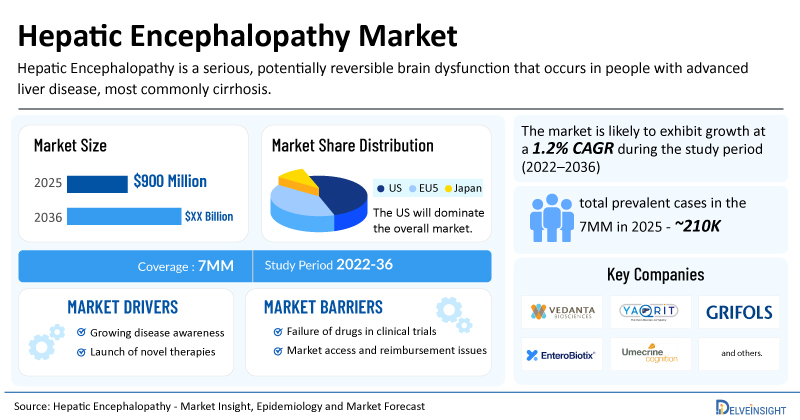
- The market size for hepatic encephalopathy was found to be USD 900 million in the 7MM in 2025.
- The United States accounted for the largest hepatic encephalopathy treatment market size in 2025, i.e 60%, compared to other major markets, including the EU4 countries (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
- The total number of prevalent cases of hepatic encephalopathy in the 7MM was nearly 210,000 cases in 2025 and is projected to increase during the forecasted period.
- Key hepatic encephalopathy companies, including Vedanta Biosciences, Yaqrit Discovery Ltd, Grifols, S.A., EnteroBiotix Limited, Umecrine Cognition AB, and others, are actively working on innovative hepatic encephalopathy drugs.
- Some of the key hepatic encephalopathy therapies in clinical trials include VE303, YAQ007, 25% IV albumin, EBX-102, GR3027, and others. These novel hepatic encephalopathy therapies are anticipated to enter the hepatic encephalopathy market in the forecast period and are expected to change the market.
- GR-3027 (Umercrine Cognition) is a potential therapy in the development of Hepatic Encephalopathy, with its expected approval by 2027 in the US.
Discover which region dominates HE market @ https://www.delveinsight.com/sample-request/hepatic-encephalopathy-market
Key Factors Driving the Growth of the Hepatic Encephalopathy Market
- Rising Hepatic Encephalopathy Prevalence: In 2025, the US accounted for approximately 200,000 diagnosed prevalent cases of hepatic encephalopathy. These cases are expected to increase during the forecast period (2026−2036) owing to increasing populations, improved diagnostic methods, advancements in medical technology allowing for better treatment, and the emergence of gene therapy and next-generation treatments, which could redefine the landscape.
- Growing Awareness and Early Diagnosis: Improved awareness among healthcare professionals and patients regarding cognitive impairment, confusion, and neurological symptoms linked to HE has led to earlier diagnosis and better treatment rates. Increased screening in cirrhosis patients is also contributing to market growth.
- Emergence of Novel Classes: The limited approved therapies for hepatic encephalopathy create a clear market opportunity for emerging treatments, including ammonia-scavengers, hyperoncotic agents, and GABAA-receptor modulating steroid antagonists (GAMSA), aimed at addressing unmet clinical needs and improving patient outcomes.
- Launch of Emerging Drugs: The dynamics of the hepatic encephalopathy market are expected to change in the coming years due to the launch of emerging therapies such as VE303 (Vedanta Biosciences), YAQ007 (Yaqrit Discovery Ltd), 25% IV albumin (Grifols, S.A.), EBX-102 (EnteroBiotix Limited), GR3027 (Umecrine Cognition AB), and others.
According to Stuti Mahajan, consulting manager at DelveInsight, the scarcity of approved treatment options for hepatic encephalopathy leaves a notable gap in the current therapeutic landscape, creating ample opportunity for emerging therapies to establish a strong market presence. Mahajan further added that this unmet need opens the door for innovative treatments with improved efficacy, safety, or differentiated mechanisms to gain a competitive advantage and drive future market growth.
Hepatic Encephalopathy Market Analysis
- Treatment for hepatic encephalopathy primarily involves DNA-directed RNA polymerase inhibitors and transcription factor inhibitors.
- Research indicates that DNA-directed RNA polymerase inhibitors may help prevent the condition by decreasing ammonia-producing gut bacteria, thereby reducing toxin accumulation that can impair brain function.
- Effective management of hepatic encephalopathy typically requires a multidisciplinary strategy that combines oral DNA-directed RNA polymerase inhibitors with supportive measures such as lifestyle changes and nutritional intervention.
- At present, only one therapy is specifically approved in the United States for hepatic encephalopathy: XIFAXAN from Bausch Health.
- Standard first-line treatment generally includes lactulose (or lactitol) along with rifaximin to lower ammonia levels, while L-ornithine L-aspartate may be used as an alternative option. In difficult or refractory cases, procedures such as shunt closure or albumin dialysis may be considered, although their survival benefits remain unclear.
- For patients with advanced disease, liver transplantation remains the only definitive treatment. Long-term care is centered on preventing recurrence through lactulose with or without rifaximin, managing triggers, and optimizing nutritional status.
- Several companies are also pursuing innovative therapies with novel mechanisms of action to address existing unmet needs and improve treatment outcomes.
- However, the late-stage pipeline for Hepatic Encephalopathy remains limited. Promising emerging candidates such as VE303 from Vedanta Biosciences, YAQ007 from Yaqrit Discovery, and GR3027 from Umecrine Cognition are anticipated to enter the hepatic encephalopathy market during the forecast period.
Learn more about the current treatment for hepatic encephalopathy @ Hepatic Encephalopathy Treatment Market
Hepatic Encephalopathy Competitive Landscape
Some of the hepatic encephalopathy drugs under development include VE303 (Vedanta Biosciences), YAQ007 (Yaqrit Discovery Ltd), 25% IV albumin (Grifols, S.A.), EBX-102 (EnteroBiotix Limited), GR3027 (Umecrine Cognition AB), and others.
Yaqrit Discovery Ltd’s YAQ007 (L-ornithine phenylacetate, L-OPA) is an oral ammonia-scavenging therapy that converts ammonia into phenylacetylglutamine, enabling its permanent elimination from the body. Yaqrit plans to begin a Phase IIb/III study of YAQ007 for the outpatient prevention of recurrent overt hepatic encephalopathy. At present, YAQ007 is also under investigation in a Phase II clinical trial for hepatic encephalopathy treatment.
Vedanta Biosciences’ VE303 is an oral, wholly owned, defined bacterial consortium consisting of eight live commensal bacterial strains, developed to prevent recurrent C. difficile infection (rCDI). It is manufactured using pure clonal cell banks to maintain standardized quality and consistency. These strains act by competing with C. difficile for nutrients while generating secondary bile acids and short-chain fatty acids, which help inhibit harmful bacterial growth, reinforce gut barrier function, and lessen intestinal inflammation. Currently, VE303 is being studied in a Phase II clinical trial for the treatment of hepatic encephalopathy.
The anticipated launch of these emerging hepatic encephalopathy therapies are poised to transform the hepatic encephalopathy market landscape in the coming years. As these cutting-edge hepatic encephalopathy therapies continue to mature and gain regulatory approval, they are expected to reshape the hepatic encephalopathy market landscape, offering new standards of care and unlocking opportunities for medical innovation and economic growth.
To know more about new drugs for hepatic encephalopathy in clinical trials, visit @ Hepatic Encephalopathy Medication
Recent Developments in the Hepatic Encephalopathy Market
- In January 2026, Bausch Health Companies Inc. announced the results of the global Phase III RED-C clinical program evaluating amorphous-rifaximin solid soluble dispersion (SSD) in adults with liver cirrhosis for the primary prevention of hepatic encephalopathy (HE). While safe and well-tolerated, neither clinical trials did not meet the primary endpoint.
- In October 2025, Biocon Pharma Limited, in partnership with Carnegie Pharmaceuticals LLC, received tentative approval from the US Food and Drug Administration (US FDA) for the ANDA for Rifaximin Tablets, 550 mg. Rifaximin tablets are a rifamycin antibacterial indicated for reducing the risk of overt hepatic encephalopathy (HE) recurrence and to treat irritable bowel syndrome with diarrhea (IBS-D) in adults.
What is Hepatic Encephalopathy?
Hepatic Encephalopathy is a serious, potentially reversible brain dysfunction that occurs in people with advanced liver disease, most commonly cirrhosis. When the liver is unable to filter toxins from the blood effectively, substances like ammonia accumulate and travel to the brain, disrupting normal neurological function. This can lead to a wide range of symptoms, from mild cognitive changes such as confusion, poor concentration, and sleep disturbances to more severe manifestations like disorientation, abnormal behavior, and even coma in advanced cases. Hepatic encephalopathy is often triggered or worsened by factors such as infections, gastrointestinal bleeding, dehydration, or constipation. Early recognition and treatment are important, as the condition can often be managed and partially reversed by addressing the underlying cause and reducing toxin buildup.
Hepatic Encephalopathy Epidemiology Segmentation
The hepatic encephalopathy epidemiology section provides insights into the historical and current V patient pool and forecasted trends for the leading markets. In the US, a consistently higher number of cases are projected to be reported in the late-onset group. The number of cases of CHE and OHE were nearly 125,000 and 80,000, respectively, in 2025.
The hepatic encephalopathy market report proffers epidemiological analysis for the study period 2022–2036 in the leading markets segmented into:
- Total Diagnosed Prevalent Cases of Hepatic Encephalopathy
- Gender-specific Cases of Hepatic Encephalopathy
- Age-specific Cases of Hepatic Encephalopathy
- Type-specific Cases of Hepatic Encephalopathy
- Grade-Specific Cases of Hepatic Encephalopathy
Download the report to understand emerging therapies in HE pipeline @ Hepatic Encephalopathy Treatment Options
| Hepatic Encephalopathy Market Report Metrics | Details |
| Study Period | 2022–2036 |
| Hepatic Encephalopathy Market Report Coverage | 7MM [The United States, the EU-4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan] |
| Hepatic Encephalopathy Market CAGR | 1.2% |
| Hepatic Encephalopathy Market Size in 2025 | USD 900 Million |
| Key Hepatic Encephalopathy Companies | Vedanta Biosciences, Yaqrit Discovery Ltd, Grifols, S.A., EnteroBiotix Limited, Umecrine Cognition AB, Bausch Health, and others |
| Key Hepatic Encephalopathy Therapies | VE303, YAQ007, 25% IV albumin, EBX-102, GR3027, XIFAXAN, and others |
Scope of the Hepatic Encephalopathy Market Report
- Hepatic Encephalopathy Therapeutic Assessment: Hepatic Encephalopathy current marketed and emerging therapies
- Hepatic Encephalopathy Market Dynamics: Conjoint Analysis of Emerging Hepatic Encephalopathy Drugs
- Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Hepatic Encephalopathy Market Unmet Needs, KOL’s views, Analyst’s views, Hepatic Encephalopathy Market Access and Reimbursement
Discover more about which companies are developing HE drugs @ Hepatic Encephalopathy Clinical Trials
Table of Contents
| 1 | Hepatic Encephalopathy Market Key Insights |
| 2 | Hepatic Encephalopathy Market Report Introduction |
| 3 | Executive Summary |
| 4 | Key Events |
| 5 | Epidemiology and Market Forecast Methodology |
| 6 | Hepatic Encephalopathy Market Overview at a Glance |
| 6.1 | Market Share (%) Distribution of Hepatic Encephalopathy by Therapies in 2025 |
| 6.2 | Market Share (%) Distribution of Hepatic Encephalopathy by Therapies in 2036 |
| 7 | Hepatic Encephalopathy: Overview |
| 7.1 | Introduction |
| 7.2 | Signs and Symptoms |
| 7.3 | Pathophysiology |
| 7.4 | Etiology |
| 7.5 | Classification of Hepatic Encephalopathy (HE) |
| 8 | Diagnosis |
| 9 | Treatment & Management |
| 10 | Epidemiology and Patient Population |
| 10.1 | Key Findings |
| 10.2 | Assumptions and Rationale |
| 10.3 | Epidemiology: 7MM |
| 10.3.1 | Total Prevalent Cases of Hepatic Encephalopathy in the 7MM |
| 10.3.2 | Gender-Specific Prevalent Cases of Hepatic Encephalopathy in the 7MM |
| 10.3.3 | Age-specific Prevalent Cases of Hepatic Encephalopathy in the 7MM |
| 10.3.4 | Type-specific Prevalent Cases of Hepatic Encephalopathy in the 7MM |
| 10.3.5 | Grade-specific Prevalent Cases of Hepatic Encephalopathy in the 7MM |
| 10.4 | The United States |
| 10.4.1 | Total Prevalent Cases of Hepatic Encephalopathy in the United States |
| 10.4.2 | Gender-specific Prevalent Cases of Hepatic Encephalopathy in the United States |
| 10.4.3 | Age-specific Prevalent Cases of Hepatic Encephalopathy in the United States |
| 10.4.4 | Type-specific Prevalent Cases of Hepatic Encephalopathy in the United States |
| 10.4.5 | Grade-Specific Prevalent Cases of Hepatic Encephalopathy in the United States |
| 10.5 | EU4 and the UK |
| 10.6 | Japan |
| 11 | Patient Journey |
| 12 | Marketed Hepatic Encephalopathy Drugs |
| 12.1 | Key Cross Competition |
| 12.2 | Xifaxan (Rifaximin): Bausch Health |
| 12.2.1 | Drug Description |
| 12.2.2 | Regulatory milestones |
| 12.2.3 | Other Development Activities |
| 12.2.4 | Summary of Pivotal Trial |
| 12.2.5 | Clinical Development |
| 12.2.5.1 | Clinical trials information |
| 12.2.6 | Safety and Efficacy |
| 12.2.7 | Analyst Views |
| 13 | Emerging Hepatic Encephalopathy Drugs |
| 13.1 | Key Cross Competition |
| 13.2 | VE303: Vedanta Biosciences |
| 13.2.1 | Product Description |
| 13.2.2 | Other Development Activities |
| 13.2.3 | Clinical Development |
| 13.2.3.1 | Clinical Trials Information |
| 13.2.4 | Safety and efficacy |
| 13.2.5 | Analyst Views |
| 13.3 | YAQ007: Yaqrit Discovery Ltd |
| 13.4 | 25% IV albumin: Grifols, S.A. |
| 13.5 | EBX-102: EnteroBiotix Limited |
| 13.6 | GR3027: Umecrine Cognition AB |
| 14 | Hepatic Encephalopathy: Seven Major Market Analysis |
| 14.1 | Key Findings |
| 14.2 | Hepatic Encephalopathy Market Outlook |
| 14.3 | Conjoint Analysis |
| 14.4 | Key Hepatic Encephalopathy Market Forecast Assumptions |
| 14.5 | The 7MM Hepatic Encephalopathy Market Size |
| 14.5.1 | Total Market Size of Hepatic Encephalopathy in the 7MM |
| 14.5.2 | Total Market Size of Hepatic Encephalopathy by therapies in the 7MM |
| 14.6 | The United States Hepatic Encephalopathy Market |
| 14.6.1 | Total Market Size of Hepatic Encephalopathy in the United States |
| 14.6.2 | Total Market Size of Hepatic Encephalopathy by Therapies in the United States |
| 14.7 | EU4 and the UK Hepatic Encephalopathy Market |
| 14.8 | Japan Hepatic Encephalopathy Market |
| 15 | Hepatic Encephalopathy Market Unmet Needs |
| 16 | Hepatic Encephalopathy Market SWOT Analysis |
| 17 | KOL Views on Hepatic Encephalopathy |
| 18 | Hepatic Encephalopathy Market Access and Reimbursement |
| 18.1 | The United States |
| 18.2 | EU4 and the UK |
| 18.3 | Japan |
| 18.4 | Summary and Comparison of Market Access and Pricing Policy Developments in 2025 |
| 18.5 | Market Access and Reimbursement of Hepatic Encephalopathy Therapies |
| 19 | Bibliography |
Related Reports
Hepatic Encephalopathy Clinical Trial Analysis Pipeline
Hepatic Encephalopathy Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key hepatic encephalopathy companies, including Vedanta Biosciences, Yaqrit Discovery Ltd, Grifols, S.A., EnteroBiotix Limited, Umecrine Cognition AB, Bausch Health, and others.
Overt Hepatic Encephalopathy Market
Overt Hepatic Encephalopathy Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key overt hepatic encephalopathy companies, including Salix Pharmaceuticals, Bausch Health, Axcella Health, Inc., Umecrine Cognition, Mallinckrodt Therapeutics, Vedanta Biosciences, Patricia Bloom, and others.
Overt Hepatic Encephalopathy Clinical Trial Analysis Pipeline
Overt Hepatic Encephalopathy Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key overt hepatic encephalopathy companies, including Salix Pharmaceuticals, Bausch Health, Axcella Health, Inc., Umecrine Cognition, Mallinckrodt Therapeutics, Vedanta Biosciences, Patricia Bloom, and others.
Crohn’s Disease Market
Crohn’s Disease Market Insights, Epidemiology, and Market Forecast – 2036 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key Crohn’s disease companies, including RedHill Biopharma, Merck, Teva Pharmaceutical, Pfizer, Agomab Therapeutics, Sanofi, Medibiofarma, Eli Lilly, NImmune, Avobis Bio, Abivax, Roche, Telavant, Mesoblast, Takeda, AstraZeneca, and others.
Cushing’s Disease Market
Cushing’s Disease Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key Cushing’s disease Market companies, including Corcept Therapeutics, Sparrow Pharmaceuticals, Crinetics Pharmaceuticals, H. Lundbeck, Stero Therapeutics, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur info@delveinsight.com +14699457679

Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. Publication takes no editorial responsibility for the same.