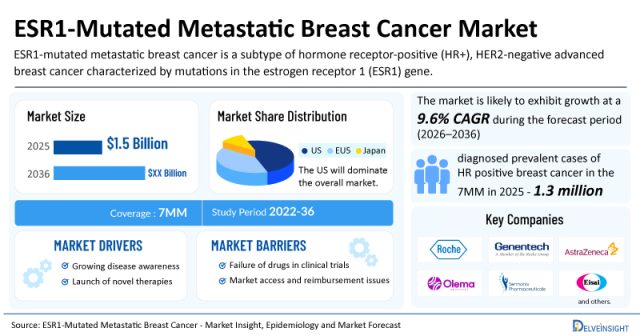
The rheumatoid arthritis clinical trial analysis report delivers important insights into ongoing research of 80+ pipeline rheumatoid arthritis drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
New York, USA, April 28, 2026 (GLOBE NEWSWIRE) — Rheumatoid Arthritis Clinical Trial Pipeline: DelveInsight Highlights Major Advances, Transformative Therapies, and 75+ Leading Players Wheeling the Therapeutics Landscape
The rheumatoid arthritis clinical trial analysis report delivers important insights into ongoing research of 80+ pipeline rheumatoid arthritis drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
DelveInsight’s ‘Rheumatoid Arthritis Pipeline Insight 2026’ report provides comprehensive global coverage of pipeline therapies for rheumatoid arthritis across various stages of clinical development. The report offers an in-depth analysis of key trends, emerging therapies, and competitive landscape dynamics, highlighting the strategies of major pharmaceutical companies to advance the pipeline and capitalize on future growth opportunities. In addition, it includes critical insights into clinical trial benchmarking, partnering and licensing activities, and regulatory pathways involving the FDA and EMA, enabling stakeholders to make informed decisions and optimize development strategies within the rheumatoid arthritis domain.
Rheumatoid Arthritis Clinical Trial Analysis Summary
- DelveInsight’s rheumatoid arthritis pipeline report depicts a robust space with 75+ active players working to develop 80+ pipeline rheumatoid arthritis drugs.
- Key rheumatoid arthritis companies, such as Lynk Pharmaceuticals, Simcere Pharmaceutical, mAbxience, SynAct Pharma, Revolo Biotherapeutics, Eli Lilly and Company, IMBiologics, Rise Therapeutics, Sonoma Biotherapeutics, DeepCure, Xencor, Inc., AbbVie, SciRhom GmbH, Novartis Pharmaceuticals, AstraZeneca, Bristol-Myers Squibb, Lifordi Immunotherapeutics, Inc., Gilead Sciences, XBiotech, Inc., Candid Therapeutics, Hangzhou Highlightll Pharmaceutical Co., Ltd., and others, are evaluating new rheumatoid arthritis drugs to improve the treatment landscape.
- Promising pipeline rheumatoid arthritis therapies, such as LNK01001, MB04, AP1189, IRL-201805, Ocadusertib, IMB 101, R-2487, SBT-77-7101, DC-9476, XmAb13676, Lutikizumab, SR-878, PIT565, AZD1163, BMS-986454, BMS-986528, LFD-200, GS-0151, Natrunix, CND261, TLL-018, and others, are in different phases of rheumatoid arthritis clinical trials.
- Approximately 25+ rheumatoid arthritis drugs are in the late stages of development.
- Notable MoAs in rheumatoid arthritis clinical trials include Janus kinase 1 inhibitor, Tumour necrosis factor inhibitor, Melanocortin type 1/3 receptor agonists, Immunomodulators, RIPK1 protein inhibitor, OX40 ligand inhibitors; Tumour necrosis factor alpha inhibitor, Regulatory T-lymphocyte stimulants, T lymphocyte replacements, BRD4 protein inhibitors, and others.
Request a sample and discover the recent advances in rheumatoid arthritis drug development @ https://www.delveinsight.com/sample-request/rheumatoid-arthritis-ra-pipeline-insight
What is Rheumatoid Arthritis?
Rheumatoid arthritis is a chronic autoimmune disorder in which the body’s immune system mistakenly attacks the lining of the joints, leading to inflammation, pain, swelling, and stiffness, often affecting the hands, wrists, and knees symmetrically. Over time, persistent inflammation can cause joint damage, deformity, and loss of function if left untreated. Rheumatoid arthritis is not just limited to joints; it can also impact other organs such as the lungs, heart, and eyes. The exact cause is unknown, but a combination of genetic predisposition and environmental triggers is believed to play a role. Early diagnosis and treatment, typically involving disease-modifying antirheumatic drugs (DMARDs) and biologics, are crucial to controlling disease progression and improving quality of life.

Find out more about rheumatoid arthritis drug development @ Rheumatoid Arthritis Treatment
A snapshot of the Pipeline Rheumatoid Arthritis Drugs mentioned in the report:
| Drugs | Company | Phase | MoA | RoA |
| LNK01001 | Lynk Pharmaceuticals/Simcere Pharmaceutical | III | Janus kinase 1 inhibitor | Oral |
| MB04 | mAbxience | III | Tumour necrosis factor inhibitor | Subcutaneous |
| AP1189 | SynAct Pharma | II | Melanocortin type 1/3 receptor agonists | Oral |
| IRL-201805 | Revolo Biotherapeutics | II | Immunomodulators | IV infusion or IV bolus |
| Ocadusertib | Eli Lilly and Company | II | RIPK1 protein inhibitor | Oral |
| IMB 101 | IMBiologics | I | OX40 ligand inhibitors; Tumour necrosis factor alpha inhibitor | Intravenous |
| R-2487 | Rise Therapeutics | I | Regulatory T-lymphocyte stimulants | Oral |
| SBT-77-7101 | Sonoma Biotherapeutics | I | T lymphocyte replacements | Single infusion |
| DC-9476 | DeepCure | Preclinical | BRD4 protein inhibitors | Oral |
Learn more about the emerging rheumatoid arthritis therapies @ Rheumatoid Arthritis Clinical Trials
According to Stuti Mahajan, consulting manager, the rheumatoid arthritis pipeline is robust and innovation-driven, with over 80 therapies in development targeting novel pathways beyond traditional TNF inhibition, including JAK inhibitors, BTK inhibitors, and cell-based immunotherapies. A clear shift toward precision immunology and mechanism diversification, which is expected to improve outcomes in patients with inadequate response to existing biologics. Additionally, Mahajan added, increasing competition from biosimilars and next-generation oral agents is likely to intensify pricing pressure, pushing companies to demonstrate superior efficacy, safety, or convenience. Overall, the RA market is viewed as highly competitive but opportunity-rich, with differentiation through novel targets and improved patient stratification expected to drive future growth.
Recent Developments in Rheumatoid Arthritis Treatment Space
- In January 2026, Lynk Pharmaceuticals Co., Ltd., a clinical-stage innovative drug development company focused on developing innovative therapies for immune and inflammatory diseases, announced positive topline results from its Phase III clinical trial evaluating zemprocitinib (LNK01001) in patients with moderate to severe active rheumatoid arthritis (RA). The study met its primary and key secondary efficacy endpoints, demonstrating statistically significant improvements versus placebo (P < 0.0001), with a favorable safety and tolerability profile.
- In October 2025, RheumaGen, Inc. and SiVEC Biotechnologies, Inc. announced a licensing and joint development agreement for a breakthrough class of gene-editing therapies designed to cure autoimmune diseases. The partnership will utilize SiVEC’s BactPac™ drug delivery platform to streamline manufacturing and increase the scalability of RheumaGen’s new class of human leukocyte antigen (HLA) gene-editing therapies for common autoimmune diseases; importantly, the partnership also establishes an in vivo program that will be co-developed by the companies.
- In March 2026, T-cell engager company Kali Therapeutics Inc. licensed its lead product, CD19/BCMA/CD3 trispecific antibody KT-501, to Sanofi SA in a global deal that could be worth more than $1.2 billion. The San Mateo, Calif.-based biotech first emerged in 2024 and moved KT-501 into a phase Ia trial last week to treat adults with rheumatoid arthritis.
- In January 2026, SetPoint Medical, a company dedicated to developing therapies for people living with chronic autoimmune diseases, announced the publication of data from the RESET-RA study in Nature Medicine. The pivotal, double-blind, randomized, sham-controlled study evaluated the safety and efficacy of the SetPoint System, an implantable vagus nerve-targeted neuroimmune modulation device for adults living with moderately-to-severely active rheumatoid arthritis (RA) who are not adequately managed by, or cannot tolerate, existing advanced RA therapies, such as biological and targeted synthetic disease-modifying anti-rheumatic drugs (DMARDs).
- In October 2025, Artiva Biotherapeutics, Inc. announced that the U.S. Food and Drug Administration (FDA) had granted Fast Track Designation to AlloNK® (also known as AB-101) for the treatment of refractory rheumatoid arthritis (RA) in combination with rituximab and that the Company has prioritized refractory RA as the program’s lead indication.
- In October 2025, Imaging technology company Adaptix began live imaging trials as part of a research program at the University of Exeter. Patients suspected of having rheumatoid arthritis will be the first in the United Kingdom to use Adaptix Ortho350 technology to help diagnose and track the progression of the disease in their hands.
- In October 2025, EVOQ Therapeutics, Inc. announced a Collaboration and License Agreement with Sanofi, an R&D driven, AI-powered biopharma company, committed to improving people’s lives and delivering compelling growth. EVOQ’s proprietary breakthrough NanoDisc technology restores the body’s natural immune tolerance pathways and opens the door for a wide range of disease-specific, curative therapies for multiple autoimmune diseases like celiac disease, type 1 diabetes, MOG (myelin oligodendrocyte glycoprotein) antibody disease, rheumatoid arthritis and lupus.
Scope of the Rheumatoid Arthritis Pipeline Report
- Coverage: Global
- Rheumatoid Arthritis Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
- Rheumatoid Arthritis Therapeutic Assessment By Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
- Rheumatoid Arthritis Therapeutics Assessment By Route of Administration: Oral, Intravenous, Subcutaneous, Parenteral, Topical
- Rheumatoid Arthritis Therapeutics Assessment By Molecule Type: Recombinant fusion proteins, Small molecule, Monoclonal antibody, Peptide, Polymer, Gene therapy
- Rheumatoid Arthritis Therapeutics Assessment By Mechanism of Action: Janus kinase 1 inhibitor, Tumour necrosis factor inhibitor, Melanocortin type 1/3 receptor agonists, Immunomodulators, RIPK1 protein inhibitor, OX40 ligand inhibitors; Tumour necrosis factor alpha inhibitor, Regulatory T-lymphocyte stimulants, T lymphocyte replacements, BRD4 protein inhibitors, and others
- Key Rheumatoid Arthritis Companies: Lynk Pharmaceuticals, Simcere Pharmaceutical, mAbxience, SynAct Pharma, Revolo Biotherapeutics, Eli Lilly and Company, IMBiologics, Rise Therapeutics, Sonoma Biotherapeutics, DeepCure, Xencor, Inc., AbbVie, SciRhom GmbH, Novartis Pharmaceuticals, AstraZeneca, Bristol-Myers Squibb, Lifordi Immunotherapeutics, Inc., Gilead Sciences, XBiotech, Inc., Candid Therapeutics, Hangzhou Highlightll Pharmaceutical Co., Ltd and others
- Key Rheumatoid Arthritis Pipeline Therapies: LNK01001, MB04, AP1189, IRL-201805, Ocadusertib, IMB 101, R-2487, SBT-77-7101, DC-9476, XmAb13676, Lutikizumab, SR-878, PIT565, AZD1163, BMS-986454, BMS-986528, LFD-200, GS-0151, Natrunix, CND261, TLL-018 and others.
Dive deep into rich insights for new rheumatoid arthritis treatments, visit @ Rheumatoid Arthritis Drugs
Table of Contents
| 1. | Rheumatoid Arthritis Pipeline Report Introduction |
| 2. | Rheumatoid Arthritis Pipeline Report Executive Summary |
| 3. | Rheumatoid Arthritis Pipeline: Overview |
| 4. | Analytical Perspective In-depth Commercial Assessment |
| 5. | Rheumatoid Arthritis Clinical Trial Therapeutics |
| 6. | Rheumatoid Arthritis Pipeline: Late-Stage Products (Pre-registration) |
| 7. | Rheumatoid Arthritis Pipeline: Late-Stage Products (Phase III) |
| 8. | Rheumatoid Arthritis Pipeline: Mid-Stage Products (Phase II) |
| 9. | Rheumatoid Arthritis Pipeline: Early-Stage Products (Phase I) |
| 10. | Rheumatoid Arthritis Pipeline Therapeutics Assessment |
| 11. | Inactive Products in the Rheumatoid Arthritis Pipeline |
| 12. | Company-University Collaborations (Licensing/Partnering) Analysis |
| 13. | Key Companies |
| 14. | Key Products in the Rheumatoid Arthritis Pipeline |
| 15. | Unmet Needs |
| 16. | Market Drivers and Barriers |
| 17. | Future Perspectives and Conclusion |
| 18. | Analyst Views |
| 19. | Appendix |
For further information on the rheumatoid arthritis cure research, reach out @ Medication for Rheumatoid Arthritis Treatment
Related Reports
Rheumatoid Arthritis Market
Rheumatoid Arthritis Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key rheumatoid arthritis companies, including Cyxone, Taiho Pharmaceutical, AstraZeneca, Eli Lilly and Company, Rise Therapeutics, TNF Pharmaceuticals, Incyte, UCB, and others.
JAK Inhibitors Market
JAK Inhibitors Market Size, Target Population, Competitive Landscape, and Market Forecast – 2034 report delivers an in-depth understanding of the market trends, market drivers, market barriers, and key JAK inhibitors companies, including Pfizer, AbbVie, Galapagos, Sierra Oncology, Theravance Biopharma, Dizal Pharmaceutical, Aclaris Therapeutics, Celon Pharma, Incyte Corporation, Gilead Sciences, Reistone Biopharma, Jiangsu Hengrui Medicine Co., MaxiNovel Pharmaceuticals, and others.
Juvenile Rheumatoid Arthritis Market
Juvenile Rheumatoid Arthritis Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key juvenile rheumatoid arthritis companies, including Eli Lilly and Company, Novartis, Sanofi, Regeneron, AbbVie, GeneScience Pharmaceuticals, Cerecor, Bio-Thera Solutions, Mycenax Biotech, Boehringer Ingelheim, and others.
Psoriatic Arthritis Market
Psoriatic Arthritis Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key PsA companies, including UCB Biopharma, Sun Pharmaceutical Industries Limited, BMS, Affibody AB, Janssen Biotech, Amgen, Eli Lilly and Company, AbbVie, Pfizer, and others.
Juvenile Idiopathic Arthritis Market
Juvenile Idiopathic Arthritis Market Insights, Epidemiology, and Market Forecast – 2036 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key juvenile idiopathic arthritis companies, including Bristol-Myers Squibb, UCB Biopharma, Novartis, AbbVie, Sanofi, Regeneron Pharmaceuticals, Janssen Pharmaceutical, Eli Lilly, Sobi, Pfizer, and others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.
CONTACT: Contact Us Shruti Thakur info@delveinsight.com +14699457679

Disclaimer: The above press release comes to you under an arrangement with GlobeNewswire. Publication takes no editorial responsibility for the same.